Capture trial data from paper case report forms and electronic CRFs (eCRFs) with eClinical
eClinical is a data capture system that combines traditional paper case report forms with electronic data capture (EDC) to enable you to process high volumes of forms with clinical efficiency, without compromising accuracy, reliability or audibility.
Speak to an expert
Request callbackRequest a callback
[contact-form-7 id="114" title="Contact form"]
As new drug and product development can take many years and cost millions, you cannot underestimate the importance of your form processing and data capture system to ensure everything is done right the first time.
However big or small the trial, it is prone to human error. As many involve a large number of trial patients and investigator sites, you have to be able to rely on your data collection processes. The simplicity of eClinical is such that any member of your clinical trials team with basic IT literacy skills can be trained up to take responsibility for designing data capture forms and system administration.
Walkthrough the steps involved to get better grasp of what it can do for you.
Design
attractive and functional questionnaires, surveys and case report forms with the easy to use CRF designer to meet trial protocol and standard operating procedures (SOP)
Capture
patient data whatever the method of delivery (fax, paper, web or eMail) for maximum efficiency
Extract
data using OCR, ICR, OMR, barcode, and logo recognition
Verify
the data to ensure only accurate results are captured
Export
data to the Trial Master File (TMF), clinical trial management software (CTMS) or related databases and applications for true integration
Archive
and retrieve forms securely and conveniently for total accountability
With the optional eForm module, eClinical can be used as a hybrid paper and electronic data capture (EDC) system. The system allows paper surveys and questionnaires to be exported from the CRF Designer as dynamic PDF or HTML forms for end-users to complete on-screen and submit electronically back to the verification module.
We also provide stand-alone EDC systems that simplify compliance with ICH GCP E6 R2 standards and other regulations such as 21CFR Part 11 with online forms, web interfaces, audit trails, access control, version tracking, electronic signatures and more.
Benefits
Consistent eCRF/CRF design
A WYSIWYG form designer allows you to create, configure and deploy forms (e.g. CRFs, PROs, patient follow-up and questionnaires) with re-usable fields, sections and design elements.
Replicate existing forms to accelerate study build
Design new paper questionnaires or capture your existing CRF designs by overlaying data entry fields on top of your current structure.
Efficiently collect information
Capture clinical data from multiple sources (MFPs, scanners, fax, eMail, web and smartphones) in both centralised and distributed capture environments.
Improve accuracy and lower risk
eClinical captures your data with clearly defined and consistent rules with non-compliant data flagged for human verification.
Archiving clinical trials data
Once content is verified, it can be exported into your TMF or eStore for archiving and retrieval. Alternatively, it can be exported to different formats including Excel, CSV, XML, SQL or SPSS.
FDA 21 CFR Part 11 compliant
eClinical can be used as part of an FDA 21 CFR Part 11 compliant system to streamline clinical trials processes and remain compliant.
Migrate from paper to electronic data capture (EDC)
eClinical allows paper case report forms to be exported from the CRF Designer as dynamic PDF or HTML forms for end-users to complete on-screen and submit electronically back to the verification module.
Improve productivity
Allow your trial team to focus on more productive tasks rather than time-consuming document sorting and manual data entry.
Reduce costs
You can save on postage and transportation, scanning bureaus and end your need for expensive storage space.
Time savings
eClinical reduces time spent on study build, medical data entry, and query management, resulting in accelerated data capture.
No lost paperwork with on-site scanning
Paper questionnaires and patient surveys are scanned by the site with a digital copy immediately available and stored in records management software.
Reduce implementation time
eClinical accelerates the time needed to build and release a new study, which can have a significant benefit in reducing your time to market.
Reduce data entry errors
Reduce data entry errors by extracting data from scanned documents using OCR, ICR, OMR, and barcode technologies. Only invalid or unrecognisable fields are shown to human operators.
Built on a solid framework
eClinical is not just an off-the-shelf data capture system; it is bespoke and tailored from off-the-shelf components. This means you get a solution that does exactly what you need, is not bloated with extra functionality and you get peace of mind that your mission critical application is built on a solid and well supported framework.
UK data centres
Our data centres are UK-based and fully compliant with UK GDPR.
If you prefer, we can host your EDC system within the Microsoft Azure global infrastructure, allowing you to keep sensitive data in any region and comply with relevant legislation.
York Teaching Hospital NHS Trust
“With eForms, the accuracy and timeliness of data collection has improved immeasurably. This was not possible with paper and means we can begin analysis much quicker to establish if the FIT can accurately detect colorectal cancer in patients”
Paper case report forms (pCRFs) library


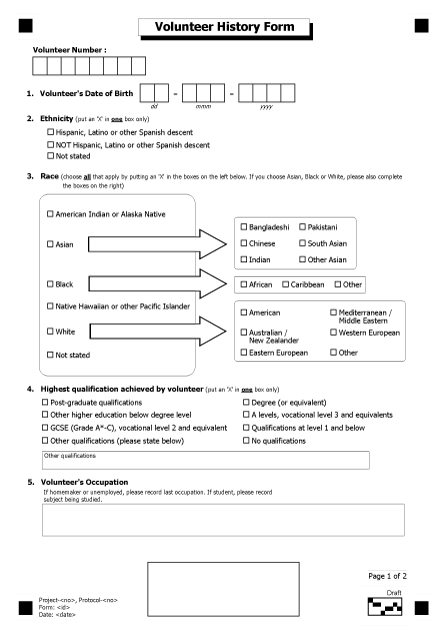





North Cumbria University NHS Hospitals Trust



London School of Hygiene and Tropical Medicine / MenAfricar

London School of Hygiene and Tropical Medicine / MenAfricar

Case studies
Contact us
eClinical is a powerful clinical data management system for hybrid paper and electronic data capture in clinical trials.
Alternatively, our stand-alone EDC platform includes online forms, web interfaces, audit trails, access control, version tracking, and electronic signatures to simplify compliance with ICH GCP E6 R2 standards and other regulations such as 21CFR Part 11.
Call +44 (0)3300 100 000 or complete this form and we will contact you to discuss your requirements.


















