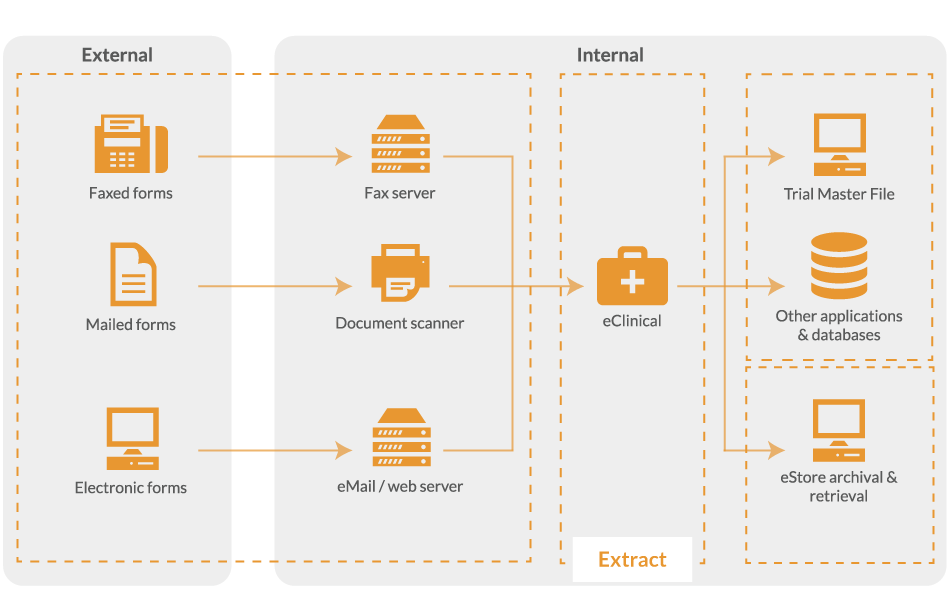
Clinical data extraction
Once document scanning is complete, the images are uploaded for image processing, classification and clinical data extraction.
Image processing and data classification
Before data can be read, the image needs to be cleaned up. eClinical will check page size and orientation, convert colour images to black and white for best recognition results and remove image defects. Additionally, the data capture software will classify data and confirm barcode readability, check the presence of cornerstones and link pages.
Extract clinical trial data using intelligent document recognition technology
After pre-processing, hand print (ICR), machine print (OCR) and checkbox (OMR) document recognition technology will extract data from a scanned document. This will include constrained print fields, tick boxes, variable scales, comments, tick boxes and signatures.
The following intelligent document recognition technology is used for clinical data extraction from documents such as CRF's, surveys and questionnaires:
- ICR (Intelligent Character Recognition) for handwritten character recognition
- OMR (Optical Mark Recognition) for recognising non-text responses on forms such as confirming if a checkbox has been filled in
- OCR (Optical Character Recognition) for reading machine printed text
- Barcode recognition of all standard barcode types, including two-dimensional (2D) matrix barcodes
- Signature detection, to ensure forms have been authorised.
Simple rules such as alpha, numeric, dictionaries, date ranges, look-ups and mandatory fields will be checked at this stage with any unrecognised fields/characters queued for human review.
These common-sense logic rules are applied to the extracted clinical data to ensure that invalid responses are not exported (e.g. impossible to tick more than one response for a single-choice question). In such situations, eClinical will be instructed to request a manual decision. The entire process takes minutes meaning thousands of forms can be processed each day.
Are you a Clinical Trials Manager looking to extract medical data from paper forms? Talk to us today to book a demonstration.