Archiving clinical trial documents in eClinical
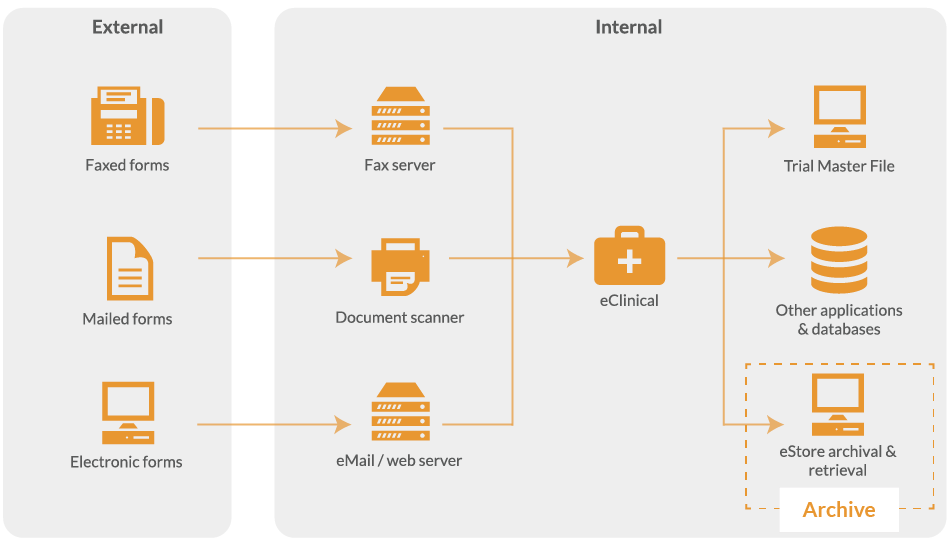
Archiving clinical trial documents is the next phase of the data capture process. Filing essential documents in a clear and timely manner greatly allows the Chief Investigator or Trial Manager to successfully manage the trial. It also assists in auditing and can help the Medicines & Healthcare Regulatory Agency (MHRA) in inspecting a trial.
At this stage, you are ready to archive trial data and store both the indexed data and the original form images for retrieval later.
If sponsors or regulators query submitted forms, you will have instant answers.
For compliance purposes, eClinical enables full automation of your disposition and retention policies to help you meet Clinical Trials Regulations. After content has been retained for the appropriate amount of time, disposition rules can automatically remove old data based on conditions you specify.
Archive and retrieve for total accountability
The extracted data is used to form an index for the optional eStore, an electronic document storage and retrieval system. This allows you to perform searches using definable criteria, across every paper form you have converted into an image.
The eStore system can also be integrated with other applications so that data collection forms can be retrieved seamlessly from within your existing applications.
If you want to learn more about archiving clinical trial documents in accordance with GCP archiving requirements, or wish to book a demonstration, please contact us.