Clinical data validation
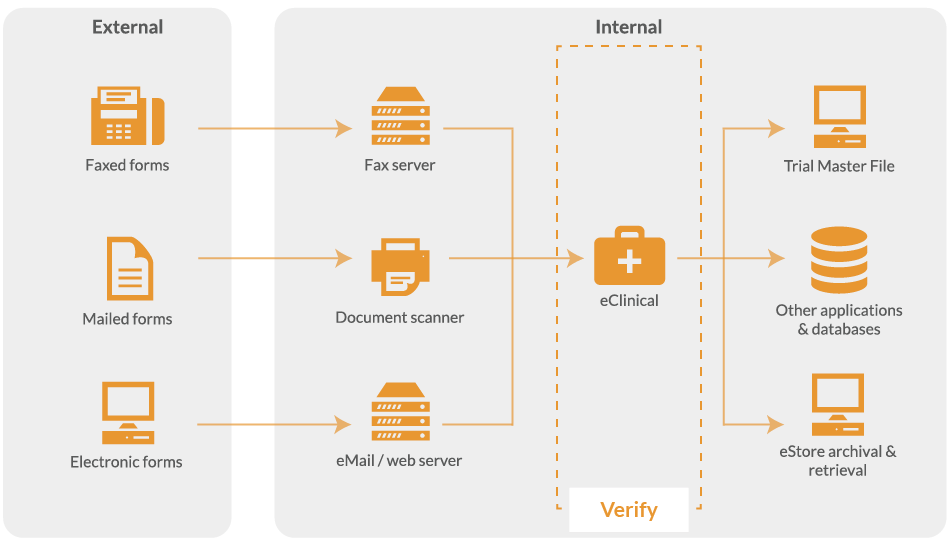
Clinical data validation and the collection of accurate trial data is essential for compliance with Standard Operating Procedures (SOP's) and Good Clinical Practice (GCP).
Consequently, you need to be able to verify data quickly and efficiently to minimise costs and improve data quality in clinical trials.
Is this name correct? Does it match the patient ID? Are the required fields completed correctly? Is the date of birth within a valid range?
Validation and verification of data for confident progression
The data capture system includes a high-speed verification interface to allow a user to verify data from a paper CRF in a few keystrokes.
You can make instant decisions on borderline characters (is this 'Jones' or 'Tones'?) at the stroke of a key.

Additionally, you can enforce rules such as numeric or alpha fields and valid ranges. Even complex rules such as age calculations and address validations against the electoral roll can be performed.
Not only does this reduce the man-hours spent capturing the trial data but it often improves accuracy. This is because human operators are focused on the fields that are unclear or invalid, rather than looking at - and keying - every single field on a form. This helps reduce the inevitable human-error from manual data entry and means the data from source documents meet Standard Operating Procedures (SOP) before it enters your trial database.
If you want to learn more about the data validation process in clinical trials and eClinical, or wish to book a demonstration, please contact us.